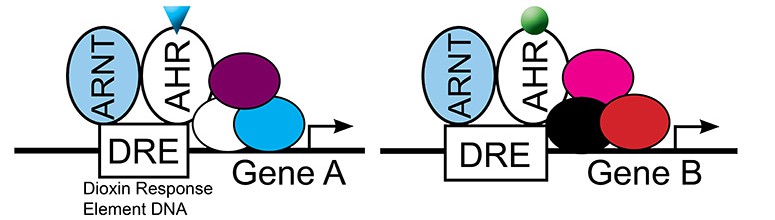
Aryl hydrocarbon receptor signaling
Exposure to byproducts of industrial combustion, such as dioxins and polycyclic aromatic hydrocarbons (PAH), causes multi-organ phenotypes including cardiotoxicity. Understanding the signaling pathways by which such compounds cause toxicity is crucial for predicting acceptable exposure levels and for reversing the effects of adverse exposure.
Dioxins and PAHs activate aryl hydrocarbon receptors (AHR), ligand-dependent transcription factors that recruit cofactors to DNA and directly regulate gene expression. However, our knowledge of AHR cofactor recruitment and regulation is limited. We designed a novel approach to identify the transcriptional cofactors by which AHR mediates cardiotoxicity and test the hypothesis that AHR ligands modulate cofactor recruitment.
Structurally distinct AHR ligands induce differential transcriptional responses, suggesting that different ligands bind AHR and recruit different cofactors, leading to differential gene expression. While the idea of ligand-dependent cofactor recruitment has been established for the estrogen receptor family of transcription factors, whether AHRs act in a similar manner is less well understood. Approximately 20 AHR cofactors have been identified, yet aside from the ARNT protein, it is not known which cofactors are required for cardiotoxicity or whether cofactors are recruited in a ligand-dependent manner.
We propose that AHR bound to the ligand TCDD recruits a different complex of transcriptional cofactors than AHR bound to other ligands, and that differential cofactor recruitment underlies ligand-dependent gene expression and cardiotoxicity.
We are identifying AHR cofactors required for TCDD-dependent cardiotoxicity. Using genetic approaches, we will test whether candidate cofactors are required for TCDD cardiotoxicity. We will also use unbiased biochemical and proteomic approaches to purify TCDD-AHR from the developing heart and identify interacting proteins.
We are also discovering ligand-dependent AHR cofactors from whole embryos and select tissues. Using zebrafish ‘knock-in’ strains with epitope tags inserted into endogenous AHR signaling genes, we are testing whether known cofactors differentially interact with endogenous AHR depending on the AHR ligand. We are also purifying AHR from zebrafish embryos and using mass spectrometry to identify cofactors bound to AHR following exposure to TCDD and other AHR ligands. Results from this project will identify a mechanism of TCDD cardiotoxicity and will provide a foundation for elucidating how the structure of AHR changes in response to differential ligand binding. Furthermore, our results will increase our understanding of the role of cofactors in endogenous AHR signaling, which may lead to improved treatment for diseases where AHR signaling is abnormal.
Publications
Souder JP, Gorelick DA. ahr2, but not ahr1a or ahr1b, is required for craniofacial and fin development and TCDD-dependent cardiotoxicity in zebrafish. Toxicological Sciences 2019
Funding
National Institute of Environmental Health Sciences 1R01ES026337-01